FAQ
 Is your question not listed below? Contact us, we are happy to answer your questions.
Is your question not listed below? Contact us, we are happy to answer your questions.
IUDs and colonization or infection with Actinomyces
Pelvic actinomycosis is an extremely rare disease that can occur in women with a long duration of intrauterine device (IUD) use. This type of abscess is usually unsuspected and, thus, diagnosed and treated surgically; however, long-duration treatment with penicillin can be completely effective. While the occurrence of actinomycosis is well documented by case reports, it is not possible to quantify the risk during IUD use. Approximately 7% of women using an IUD may have a finding of Actinomyces-like organisms on a Pap test. The prognostic significance of this finding is minimal because of the lack of sensitivity and specificity and a low positive predictive value. In the absence of symptoms, women with Actinomyces-like organisms on a Pap test do not need antimicrobial treatment or IUD removal.
More: Westhoff C. Contraception 2007 Jun;75(6 Suppl):S48-50.
Chronic endometritis in women using IUD
The intrauterine device as a factor in the etiology of pelvic inflammatory disease was associated with early forms of the device, in particular, the Dalkon Shield. The incidence of pelvic inflammatory disease is not higher in users of modern intrauterine devices than in non-users. [1-3]
Endometritis is a polymicrobial disease involving, on average, 2-3 organisms. In most cases, it arises from an ascending infection from organisms found in the normal indigenous vaginal flora. Commonly isolated organisms include Ureaplasma urealyticum, Peptostreptococcus, Gardnerella vaginalis, Bacteroides bivius, Chlamydia trachomatis and group B Streptococcus.
Diagnosis usually is based on clinical findings, as follows: fever, lower abdominal pain, foul-smelling lochia in the obstetric population, abnormal vaginal bleeding, abnormal vaginal discharge, dyspareunia (may be present in patients with pelvic inflammatory disease [PID]), dysuria (may be present in patients with PID).
Endometrial biopsy can be obtained to assess chronic endometritis. The diagnosis is based on the presence of plasma cells in the endometrial stroma upon histopathological examination. [4]
The role of endocervical cultures for diagnosis is controversial. They are not generally helpful in management, as positive results are usually the result of contamination from normal resident cervicovaginal flora. However, endocervical cultures (or DNA probe) are obtained for gonorrhea and chlamydia when appropriate.
A combination regimen of ampicillin, gentamicin, and metronidazole provides coverage against most of the organisms that are encountered in serious pelvic infections. Doxycycline should be used if Chlamydia is the cause of the endometritis.
References
- Hardeman J, Weiss BD. Intrauterine devices: an update. Am Fam Physician. 2014;89(6):445-50.
- Farley TM, Rosenberg MJ, Rowe PJ, Chen JH, Meirik O. Intrauterine devices and pelvic inflammatory disease: an international perspective. Lancet. 1992;339:785-8.
- Grimes DA. Intrauterine device and upper-genital-tract infection. Lancet. 2000;356:1013-9.
- Kasius JC, Broekmans FJ, Sie-Go DM, et al. The reliability of the histological diagnosis of endometritis in asymptomatic IVF cases: a multicenter observer study. Hum Reprod. 2012; 27:153-8.
Are copper IUDs dangerous in women with Wilson disease?
Individuals with Wilson’s disease, a disorder of copper metabolism, could develop serious and irreversible neurological and liver problems from excessive copper deposits in the tissues. What the science tells us is there is a local increase in copper levels in the lining of the uterus and in the fallopian tubes (meaning the copper IUD releases copper). That makes sense as copper affects sperm capacitation, a step the sperm need to complete so they can penetrate the egg). Therefore, for the copper IUD to work the copper has to reach the area of the reproductive tract where fertilization happens (typically the fallopian tube). However, in these studies there was no change in copper levels in the blood. One study also looked at level of ceruloplasmin, the protein that carriers copper in the blood, and it stayed the same as well. If excessive copper were getting into the blood, the body would also make more ceruloplasmin to compensate.
What is the thickness of the uterine fundus?
The thickness of the uterine fundus was studied at the department of Ob/Gyn, University Hospital Zürich, Switzerland in 100 parous and nulliparous women with normal uteri using 3D ultrasound. A fundal thickness of 12.02 ± 2.03 mm (mean ± SD) was recorded.1
These results confirm earlier measurement in parous and nulliparous women conducted in 405 parous (n=229) and nulliparous (n=176) women using 2D ultrasound. In this study a fundal thickness of 12.0 ± 2.0 mm was measured (median ± SD). The lowest value was 10 mm.2
Comment: although these studies show lowest values of ~10 mm, sometimes slightly lower values are found. In these cases insertion of the GyneFix can be done performed by slowly and gently inserting the anchor and by checking the marker afterwards. Care must also be taken to insert the anchor in the midline.
References
- Bajka et al. Ultrashall Med. 2015 June 17 (Epub ahead of print).
- Wildemeersch et al. Contraception 1992;46:471-6.
Migrations of an IUD
Migration of an IUD could occur if the IUD is too big for the uterine cavity. Many uterine cavities are extremely narrow. If not expelled, migration is caused by asymmetrical contractions of the uterus exerted on the IUD embedding the IUD in the wall of the uterus or causing even secondary perforation. If the IUD is embedded in the myometrium, operative hysteroscopy may be required for removal.
When the anchoring knot is inserted in the muscular wall of the uterus, migration of the GyneFix® is not possible as the uterine contractions have no impact on GyneFix®. Perforations of the uterus usually occur at insertion. Surgery reports showed that in most cases, the IUD was found attached to the small bowel, the omentum or mesentery.
Perforation
Perforations are more likely if no traction to the cervix is exerted. Aligning the uterus is important to allow insertion in the midline where the fundus is thickest. In cases of a pronounced uterus arcuatus (the finding of a thick fundus on ultrasound in a young woman), the inserter tube could glide to the side. Cornual insertion can be prevented by continuous traction on the cervix with a tenaculum until the anchoring is completed.
Low position of IUD
Ideally, an IUD should be positioned in the fundal region of the uterus for optimal contraception. Usually, GyneFix® is anchored in the fundus and this is the reason for its high efficacy.
Abnormal breakthrough bleeding and spotting in users of copper IUDs
Possible causes include IUD displacement, pregnancy (intrauterine or ectopic), and infection. Gynecologic disorders of the cervix or uterus (e.g., leiomyomas, polyps, endometrial hyperplasia and cancer of the endometrium) should be excluded. An endometrial biopsy using a Pipelle can be performed with the IUD in place.
Chronic endometritis is a common finding in endometrial biopsies of women who have abnormal bleeding and spotting. Antibiotic treatment may be useful as well as progestogens during the second part of the menstrual cycle in case a hormonal disturbance is suspected. The IUD should be removed if the complaints do not disappear and another method of contraception or insertion of a LNG-IUS can be considered.
Pelvic Inflammatory Disease (PID)
The risk of pelvic infection, which may lead to infertility, ectopic pregnancy, and chronic pelvic pain, remains one of the major concerns of IUD providers as well as of women. There is good scientific evidence that the risk of pelvic inflammatory disease (PID) is not increased after the first month following insertion of the IUD. Investigations by WHO showed that the risk of PID is limited to the first 20 days after insertion (Farley et al. Intrauterine devices and pelvic inflammatory disease: an international perspective. Lancet 1992;339:785–788. Many practitioners however remain concerned about PID, especially in higher risk populations. WHO suggests that the benefits of IUDs generally outweigh the risks in women of any age, whether parous or not, and that IUDs can be inserted in women younger than 20. However, WHO advises against the use of IUDs in women who have had PID in the previous 3 months. A practical solution is to test all high risk patients and to place the IUD on the same day. If the test results are positive then treatment should be administered immediately (a single oral dose of 1 g of azithromycine or 2×100 mg of oral doxycycline for 7 days). Women who develop symptoms of PID after IUD insertion can be safely treated with antibiotics without removing the IUD.
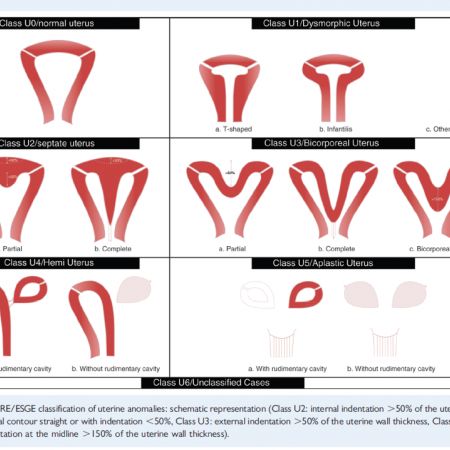
ESHRE classification of congenital anomalies of the uterus.
Infection at the anchor site
Neither infection nor adenomyosis at the anchor site have ever been reported.
Lost thread
If the tail of the GyneFix® is not visible, it can usually be found in the cervix with an alligator forceps. If the tail is curled up in the uterine cavity, I recommend to use a thin (3 mm) alligator forceps to remove the device. Usually this is successful.
Cervical stenosis
Cervical stenosis can be suspected if the “Cotton Swab Test” is negative. In this case use an os finder or a thin (3 mm) Hegar dilator to find the os and dilate up to Hegar 4 ½ - 5.
Pain control and comfort during insertion
After disinfection and gentle straightening of the uterus, a “cotton swab test” (soaked in antiseptic solution) can be done to test the tightness of the internal cervical os and to obtain information on pain sensation. If the test provokes severe pain, additional local anesthesia can be provided prior to sounding the uterus. Additional instrumentation (eg, os finder, cervical dilator), except for a sound to measure the length and flexion of the uterus, is rarely necessary if the test shows that that cavity can be entered easily.
Many believe that the use of misoprostol greatly facilitates IUD insertion. We recommend the use of 200–400 mg of misoprostol, orally or vaginally, 3 hours before IUD insertion. Others prefer to place the tablets vaginally in the posterior fornix the night before the procedure (9–12 hours before). Despite conflicting published data about the benefits of misoprostol, significant differences were found in nulliparous women between groups using misoprostol, 400 mg vaginally, 4 hours before insertion, compared to placebo with less difficulty and less moderate-to-severe pain at IUD insertion. It may be that the vaginal route could be preferable as plasma concentrations of misoprostol remain substantially higher than when administered by the oral or sublingual routes. However, most women prefer the oral route to vaginal application. If misoprostol is used for cervical priming, it is recommended that a nonsteroidal analgesic be added to reduce its prostaglandin-mediated side effects and uterine cramping. Of relevance, misoprostol could help reduce failed insertion. In a recent study among nulliparous women, 27 of 138 (19.6%) TCu380A IUD insertions were unsuccessful for emergency contraception. Some physicians claim that applying heat to the lower abdomen (using electric heating pad, or a microwave-heated cherry seed pillow) may significantly reduce painful sensations. If the patient is having severe discomfort with the insertion of the sound or requires cervical dilation, then the administration of a paracervical block or even conscious sedation (such as propofol or midazolam) can be used. However, this is rarely necessary and perhaps only in extremely anxious women.
Marker too close to serosa
Prior to insertion, the fundal thickness should be evaluated and after insertion the marker should be visualized by ultrasound and the distance measured from the serosa. Usually the marker is around 3-5 mm from the serosa (range 3-9). If the marker is very near or level with the serosa, it could be recommended to leave the IUD in place and conduct an ultrasound check after 2 weeks. If the provider elects to remove the IUD, a new insertion can be performed at once or at a later date. (see "The anchor of the frameless intrauterine device does not migrate over time: an analysis in over 300 women" in the publicationsection of this website.)
Penile pain caused by the tail of GyneFix®
Personally I trim the tail of GyneFix® about 3 cm from the cervix and then loop the tail with an alligator forceps or long pincet in the cervical canal. Most often the tail stays in place. If it comes out again, the tail will be in the fornix due to memory of the plastic suture.
Anticoagulant therapy
Patients receiving anticoagulants or having a coagulopathy may have a greater risk of menorrhagia or hypermenorrhea. They can definitely try to use GyneFix® as the majority of women using GyneFix® have no increased bleeding. LNG-IUS is indicated if women complain about increased bleeding with GyneFix®.
Abnormal bleeding
Most copper IUDs are associated with changes in uterine bleeding patterns, which may include episodes of intermenstrual bleeding and increased volume of menstrual bleeding. Counseling women about the expected changes in bleeding patterns prior to insertion may enhance adherence to the method.
Unexpected changes in bleeding patterns or changes that are not tolerable to the patient should be evaluated. Possible causes of new onset abnormal bleeding after prolonged use of IUDs include displacement of the device, pregnancy (intrauterine or ectopic), and infection, as well as gynecologic disorders of the cervix or uterus (eg, leiomyomas, polyps, endometrial cancer).
An endometrial biopsy using a Pipelle can be performed with the IUD in place. Chronic endometritis is a common finding in endometrial biopsies. A course of antibiotics may be useful. Alternatively, progestogens in the second half of the menstrual cycle may be useful if a hormonal disturbance is suspected.
Nickle allergy
A new study shows that nickel-titanium alloys in wires used in cardiovascular implants bear no increased risk for allergy sufferers. The nickel released in the long run amounted to just a few nanograms per day, which is significantly below what a human body absorbs through food on a daily basis (Posted in Medical Materials - Raw Materials by Thomas Klein on March 14, 2014).
Ectopic pregnancy
The risk of ectopic pregnancy with copper and hormonal IUDs has been extensively studied with 0.0 to 0.5/1000 women using Cu-IUDs or hormonal IUDs (Mirena) against 3.25 to 5.25/1000 women in the general population.This finding is confirmed by a meta-analysis from 16 case-control studies.Finally, there is no contraindication for the placement of an IUD in women with a history of ectopic pregnancy.
With GyneFix, over the past 20 years, ectopic pregnancy has been extremely rare. A study conducted by the World Health Organization showed a significantly lower ectopic pregnancy rate compared with the Cu-T 380A IUD.
References
- Sivin I. Dose-and age-dependent ectopic pregnancy risks with intrauterine contraception. Obstetrics and Gynecology 1991, 78) :291-8.
- Xiong X, Buekens P, Wollast E. IUD use and the risk of ectopic pregnancy: a meta-analysis of case-control studies. Contraception. 1995 Jul; 52 (1) :23-34.
Pregnancy with IUD in situ
If a pregnancy should occur with GyneFix® in situ, the device should be removed if the string is visible even though removal may increase the chances of miscarriage. If the decision is made not to remove the device or removal proves to be difficult, or if the woman elects to continue the pregnancy, she should be warned that there may be an increased risk of abortion or preterm labour and/or sepsis and she should be followed closely. A Chlamydia smear could be useful. If there is no infection, the pregnancy can go on as usual and the patient can be reassured. After the delivery look for the device in the placenta membranes. Nothing of this kind has even been reported.
Retroversion and retroflexion
Usually, the utero-cervical axis can be aligned by slight traction on the cervix. The effect of cervical traction with a tenaculum on the uterocervical angle was evaluated in 24 women (Johnson N, Bromham DR. Br J Gynaecol. 1991;98:309-12). A radio-opaque guidewire was inserted through the cervical canal into the uterine cavity and a lateral pelvic X-ray taken before and after traction.
Cervical traction in a caudal direction (force 2 N) reduced the uterocervical angle, from 75 degrees to 10 degrees. Moderate cervical traction straightens the uterus and the routine use of a tenaculum theoretically makes insertion of an intrauterine device safer.
In his clinic in Ghent, Dr. Wildemeersch organizes training sessions for doctors from inside and outside Belgium, interested in learning the anchoring technique of frameless intrauterine devices.
The clinic is specialized in providing precision intrauterine contraception to fulfill women's needs.