Products
 Dr. Wildemeersch has produced several innovative solutions in IUD & IUS contraception. These findings have been integrated in our product line, of which several are already available on the market today. We are constantly working on improving our current products and developing new innovations to improve women’s reproductive health around the world.
Dr. Wildemeersch has produced several innovative solutions in IUD & IUS contraception. These findings have been integrated in our product line, of which several are already available on the market today. We are constantly working on improving our current products and developing new innovations to improve women’s reproductive health around the world.
GyneFix
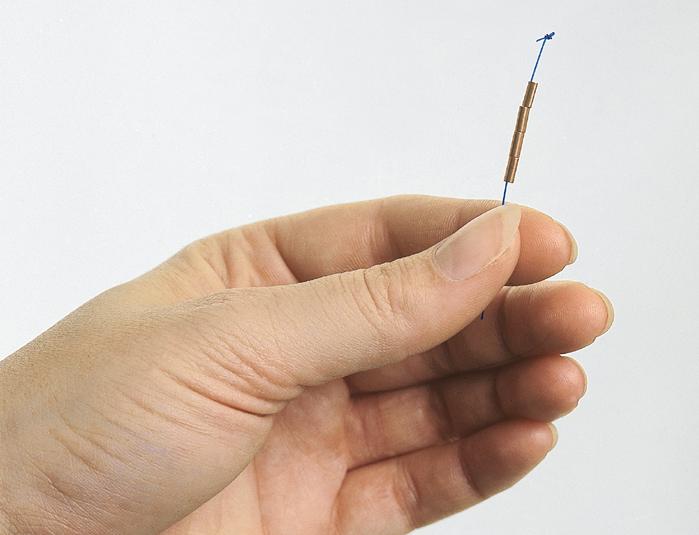
GyneFix® is a very small copper containing intrauterine device (IUD). It is suitable for insertion in all women regardless the size of their uterine cavity. The device is frameless, lacking a crossarm, is flexible and, when inserted properly, the majority of women will keep the IUD for its full lifespan of 5 years, without any pain or discomfort. The size of an IUD is closely related to the side effects it causes, so that smaller devices are better tolerated with less effect on the amount of menstrual blood loss.
Gynefix-Cesarean Section
ReLARC
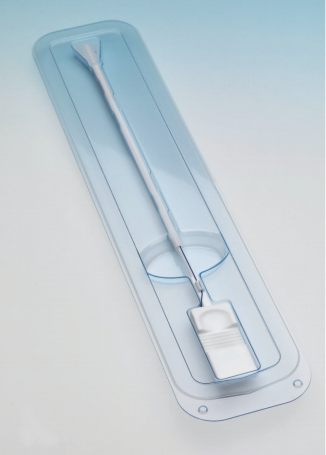
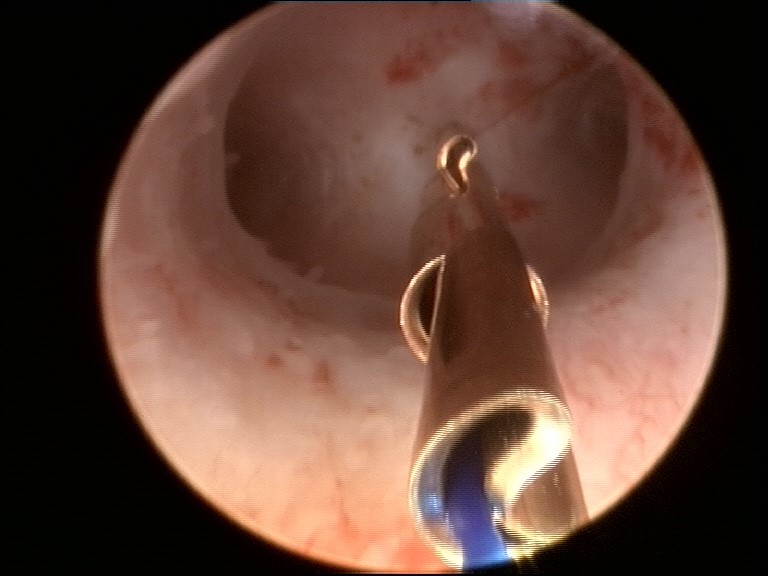 ReLARC (Reversible Long-Acting Reproductive Control is a new hysteroscopic technique for the insertion of long-term contraceptive devices. The procedure is conducted under direct visualization.
ReLARC (Reversible Long-Acting Reproductive Control is a new hysteroscopic technique for the insertion of long-term contraceptive devices. The procedure is conducted under direct visualization.
The method is simple, safe and rapid, and is suitable for application in the operating room or the office. The method is a reversible alternative to more complex insertion techniques for hysteroscopic irreversible intratubal contraception. It is the first hysteroscopically inserted system suitable for long-term intrauterine contraception up to 10 years.
Femilis Cu-T 300
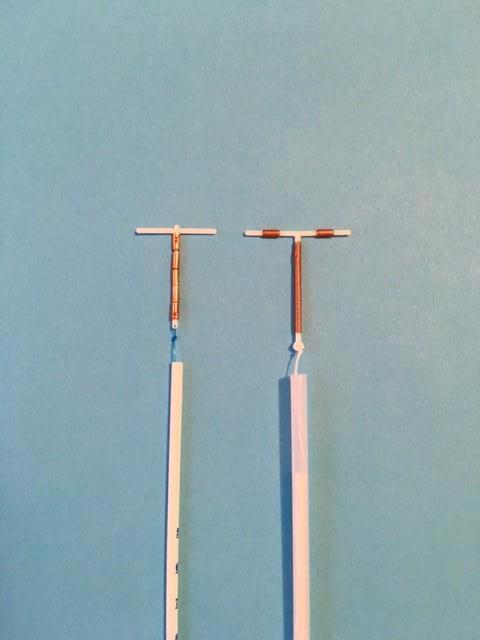 It is a T-shaped copper-releasing IUD (left) derived from the T-shaped Femilis® LNG-IUS and the frameless copper IUD, GyneFix®. The copper cylinders are open to the endometrial environment, therefore releasing a high amount of copper ions to prevent fertilization. The IUD is significantly smaller that the T-shaped® IUD, with shorter retention arm as well as stem. The device is therefore more suitable for the smaller uterine cavities of nulliparous women. Insertion is easily and safely accomplished by pushing the device in the uterine cavity (push-in technique). Provider training is therefore limited. The inserter tube is only 3 mm in diameter.
It is a T-shaped copper-releasing IUD (left) derived from the T-shaped Femilis® LNG-IUS and the frameless copper IUD, GyneFix®. The copper cylinders are open to the endometrial environment, therefore releasing a high amount of copper ions to prevent fertilization. The IUD is significantly smaller that the T-shaped® IUD, with shorter retention arm as well as stem. The device is therefore more suitable for the smaller uterine cavities of nulliparous women. Insertion is easily and safely accomplished by pushing the device in the uterine cavity (push-in technique). Provider training is therefore limited. The inserter tube is only 3 mm in diameter.
Fibroplant
Femilis LNG-IUS
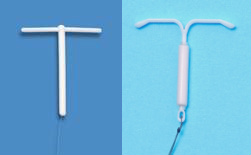 Femilis consists of a 3 cm long and a 2.4 mm wide drug delivery compartment. The vertical drug delivery rod contains 60 mg of levonorgestrel (LNG), and is covered by a rate-controlling membrane. The drug delivery compartment releases approximately 20 μg of LNG in vitro daily. The transverse retention arm has a length of either 28 mm or 24 mm to accommodate different widths of uterine cavities. Femilis® LNG-IUS is inserted using the simple and safe push-in technique, requiring minimal training. The insertion of Femilis® is apparently simpler than that required for other conventional T-shaped devices, since its arms are folded downward during the insertion process. Over 10 years of experience has been accumulated with the T-shaped LNG-IUS.
Femilis consists of a 3 cm long and a 2.4 mm wide drug delivery compartment. The vertical drug delivery rod contains 60 mg of levonorgestrel (LNG), and is covered by a rate-controlling membrane. The drug delivery compartment releases approximately 20 μg of LNG in vitro daily. The transverse retention arm has a length of either 28 mm or 24 mm to accommodate different widths of uterine cavities. Femilis® LNG-IUS is inserted using the simple and safe push-in technique, requiring minimal training. The insertion of Femilis® is apparently simpler than that required for other conventional T-shaped devices, since its arms are folded downward during the insertion process. Over 10 years of experience has been accumulated with the T-shaped LNG-IUS.
Femilis Slim LNG-IUS
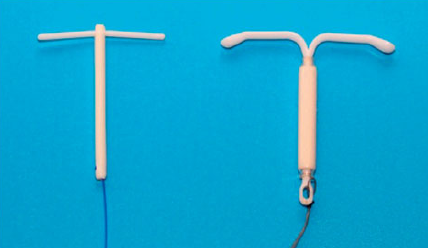 The small Femilis® Slim with average release of 14 μg per day of LNG over a 5-years has shown to be highly suitable for endometrial suppression in postmenopausal women in combination with parenteral estrogen substitution therapy. The low systemic absorption of LNG could be desirable, thus allowing for maximization of the beneficial effects of ET on organ tissues (e.g. cardiovascular tissues and breast). Use of the LNG-IUS is associated with high patient satisfaction. The risk of breast cancer may not be increased due to the very low systemic levels of the progestogen, in contrast with systemic progestogen administration.
The small Femilis® Slim with average release of 14 μg per day of LNG over a 5-years has shown to be highly suitable for endometrial suppression in postmenopausal women in combination with parenteral estrogen substitution therapy. The low systemic absorption of LNG could be desirable, thus allowing for maximization of the beneficial effects of ET on organ tissues (e.g. cardiovascular tissues and breast). Use of the LNG-IUS is associated with high patient satisfaction. The risk of breast cancer may not be increased due to the very low systemic levels of the progestogen, in contrast with systemic progestogen administration.
In his clinic in Ghent, Dr. Wildemeersch organizes training sessions for doctors from inside and outside Belgium, interested in learning the anchoring technique of frameless intrauterine devices.
The clinic is specialized in providing precision intrauterine contraception to fulfill women's needs.